Sacituzumab govitecan in ER+/HER2- metastatic breast cancer

The phase 3 trial TROPiCS-02 shows that the antibody-drug conjugate is safe and effective in heavily treated metastatic breast cancer with limited treatment options
Predictive Factors of Immunotherapy Response in Triple-Negative Breast Cancer
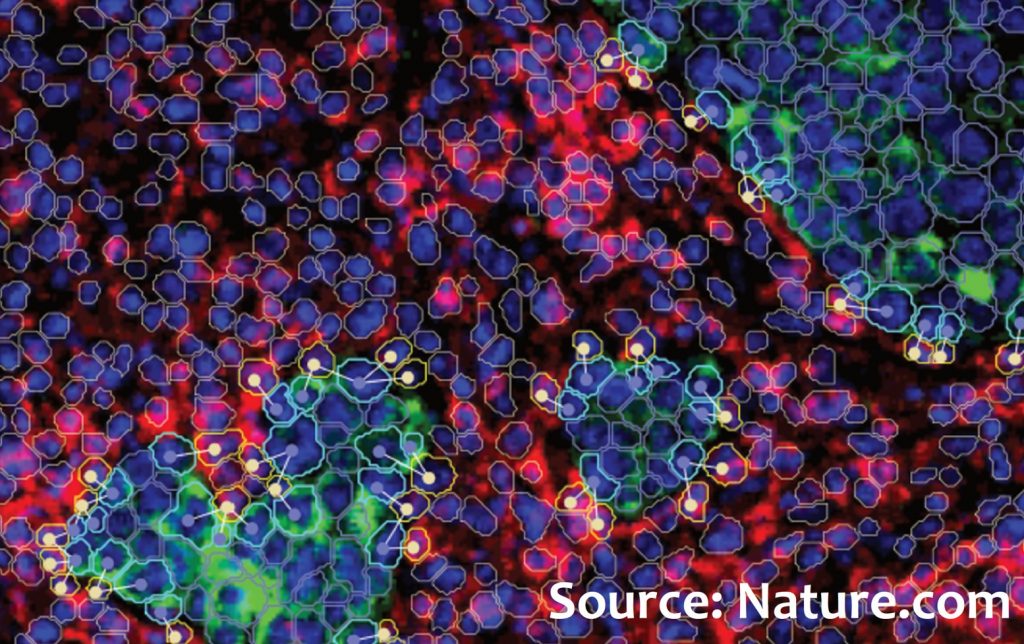
A new study by the Michelangelo Foundation, in collaboration with the CRUK Cambridge Institute, demonstrates that the spatial organization and interaction between tumour cells and various cell types critically influence the effect of immune checkpoint blockade.
Olaparib in BRCA-mutated, HER-2 negative metastatic breast cancer
Olaparib significantly prolonged progression-free survival compared to chemotherapy, particularly in first line patients.
Federated learning to predicting chemotherapy response in triple-negative breast cancer
This multicentric machine learning model improves performance of local models in predicting response to neoadjuvant chemotherapy in triple-negative breast cancer.
Fondazione Michelangelo at the San Antonio Breast Cancer Symposium

Foundation’s experts and data in key educational sessions to share knowledge on the role of neoadjuvant therapy and imaging mass cytometry in breast cancer
Triple negative breast cancer, new therapeutic options and future strategies
Lead by Michelangelo Foundation and Gianni Bonadonna Foundation’s experts, a review published on Nature Reviews – Clinical Oncology highlights the state of the art therapy in triple-negative breast cancer
HER2+ and ER+ breast cancer, promising results with trastuzumab, pertuzumab and palbociclib
NA-PHER2 trial, coordinated by Michelangelo Foundation, shows their efficacy as neoadjuvant therapy, with a relevant effect on Ki67
Triple negative breast cancer, new data from Michelangelo Foundation’s NeoTRIP trial
Atezolizumab added to nab-paclitaxel and carboplatin as neoadjuvant therapy did not increase the rate of pathological complete remission, mostly influenced by PD-L1 expression
Breast cancer, a mechanism to bypass anti-ER and anti-HER2 resistance
CDK4/6 inhibition with palbociclib, in combination with fulvestrant, trastuzumab and pertuzumab, triggers sustained senescence: new data from a Michelangelo Foundation study


